Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Komiyama, Daisuke; Amaya, Masaki
JAEA-Research 2016-013, 20 Pages, 2016/08
The boric acid in the coolant may precipitate on the fuel cladding surface in the case that the blockage of coolant flow path etc. occurs and/or the cooling of fuel becomes insufficient during a LOCA (Loss-Of-Coolant Accident) in PWRs. While there is much knowledge about the compatibility between Zircaloy-4 and boric acid under normal operation conditions, such knowledge under high temperature condition, e.g. LOCA, has not been sufficient. In this study, isothermal heating tests were carried out by using Zircaloy-4 plates with solid boric acid in various atmospheres at up to 900 C; and the stability of boric acid, the reaction between Zircaloy-4 and boric acid and the effect of solid boric acid on the high temperature oxidation behavior of Zircaloy-4 were investigated. From the results obtained, it was suggested that, if boric acid anhydride remained on the surface of Zircaloy-4, the boric acid anhydride prevented the surface of Zircaloy-4 from contacting oxidizing atmosphere and mitigated the high-temperature oxidation of Zircaloy-4. In the case that solid boric acid adhered to the surface of pre-oxidized Zircaloy-4 and they have been heated up to high temperature, it was indicated that the solid boric acid penetrated into the gaps in the pre-oxide layer and the penetrated solid boric acid mitigated the oxidation of Zircaloy-4 thereafter.
C; and the stability of boric acid, the reaction between Zircaloy-4 and boric acid and the effect of solid boric acid on the high temperature oxidation behavior of Zircaloy-4 were investigated. From the results obtained, it was suggested that, if boric acid anhydride remained on the surface of Zircaloy-4, the boric acid anhydride prevented the surface of Zircaloy-4 from contacting oxidizing atmosphere and mitigated the high-temperature oxidation of Zircaloy-4. In the case that solid boric acid adhered to the surface of pre-oxidized Zircaloy-4 and they have been heated up to high temperature, it was indicated that the solid boric acid penetrated into the gaps in the pre-oxide layer and the penetrated solid boric acid mitigated the oxidation of Zircaloy-4 thereafter.
 film formed by a hyperthermal O-atom beam at room temperature
film formed by a hyperthermal O-atom beam at room temperatureTagawa, Masahito*; Sogo, Chie*; Yokota, Kumiko*; Hachiue, Shunsuke; Yoshigoe, Akitaka; Teraoka, Yuden
Japanese Journal of Applied Physics, 44(12), p.8300 - 8304, 2005/12
Times Cited Count:5 Percentile:21.78(Physics, Applied)Si oxide layers formed on Si(001) substrates by irradiation of hyperthermal oxygen atomic beams at room temperature were analysed at the JAERI soft X-ray beamline by photoemission spectroscopy. It was found that sub-oxide components were scarcely observed in the Si oxide layers formed by the atomic oxygen beam.
Hirota, Koichi; Kojima, Takuji
Bulletin of the Chemical Society of Japan, 78(9), p.1685 - 1690, 2005/09
Times Cited Count:7 Percentile:31.71(Chemistry, Multidisciplinary)Decomposition behavior under electron-beam irradiation was examined for polychlorinated dibenzo-p-dioxin (PCDD) and polychlorinated dibenzofuran (PCDF) isomers in incinerator gases. Significant decomposition was obtained for all PCDD isomers over the investigated absorbed doses, which was resulted from oxidation reactions with OH radicals yielded by electron-beam irradiation. In the case of PCDFs, specific isomers having symmetrical structures such as 1, 2, 8, 9-, 2, 3, 7, 8-, and 3, 4, 6, 7-TeCDFs were yielded through the dechlorination of PeCDF isomers. Thermal electrons probably played a role in the dechlorination of PeCDF isomers.
Moritani, Kosuke; Okada, Michio*; Sato, Seiichi*; Goto, Seishiro*; Kasai, Toshio*; Yoshigoe, Akitaka; Teraoka, Yuden
Journal of Vacuum Science and Technology A, 22(4), p.1625 - 1630, 2004/08
Times Cited Count:25 Percentile:65.99(Materials Science, Coatings & Films)We studied the oxidation of Cu{111} surface with a hyperthermal O molecular beam (HOMB) using X-ray photoemission spectroscopy (XPS) in conjunction with a synchrotron radiation (SR) source. The efficiency of oxidation with 0.6-eV-HOMB is higher thab that with 2.3-eV-HOMB under
molecular beam (HOMB) using X-ray photoemission spectroscopy (XPS) in conjunction with a synchrotron radiation (SR) source. The efficiency of oxidation with 0.6-eV-HOMB is higher thab that with 2.3-eV-HOMB under 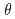
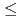 0.5ML. Ont the other hand, further oxidation occurs rather inefficiency under
0.5ML. Ont the other hand, further oxidation occurs rather inefficiency under 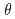
 0.5ML. In this region, efficiency of oxidation with 2.3-eV-HOMB is higher than 0.6-eV-HOMB. We found that such slow oxidation process of Cu can be interpreted in terms of a collision-induced-adsorption mechanism. These results suggest that we can control the oxidation process of Cu by using HOMB.
0.5ML. In this region, efficiency of oxidation with 2.3-eV-HOMB is higher than 0.6-eV-HOMB. We found that such slow oxidation process of Cu can be interpreted in terms of a collision-induced-adsorption mechanism. These results suggest that we can control the oxidation process of Cu by using HOMB.
Nagase, Fumihisa; Fuketa, Toyoshi
Journal of Nuclear Science and Technology, 41(7), p.723 - 730, 2004/07
Times Cited Count:45 Percentile:92.54(Nuclear Science & Technology)Experiments simulating loss-of-coolant accident (LOCA) conditions were performed to evaluate effect of pre-hydriding on thermal-shock resistance of oxidized Zircaloy-4 cladding. Artificially hydrided (400 to 600 ppm) and non-hydrided claddings were subjected to the tests. Since cladding fracture on quenching primarily depends on the oxidation amount, fracture threshold was evaluated in terms of "Equivalent Cladding Reacted (ECR)". Under axially non-restrained condition, the fracture threshold is 56% ECR and the influence of pre-hydriding is not seen. The fracture threshold is decreased by restraining the test rods on quenching, and it is more remarkable in pre-hydrided claddings than in non-hydrided claddings. Consequently, the fracture threshold was 20% ECR and 10% ECR for non-hydrided and pre-hydrided claddings, respectively, under the fully restrained condition. These results indicate possible decrease of fracture threshold of high burnup fuel claddings under LOCA conditions.
Nagase, Fumihisa; Fuketa, Toyoshi
NUREG/CP-0185, p.321 - 331, 2004/00
With a view to obtaining basic data to evaluate high burnup fuel behavior under loss of coolant accident (LOCA) conditions, a research program is being conducted at the Japan Atomic Energy Research Institute (JAERI). The program consists of integral thermal shock tests and other separate tests for oxidation rate and mechanical property of fuel claddings. Prior to the tests on irradiated claddings, the tests have been conducted on non-irradiated claddings to examine separate effects of corrosion and hydrogen absorption during reactor operation. Hydrogen effects have been especially examined because hydrogen absorption has the great impact on cladding embrittlement. The tests on irradiated claddings have recently been started and preliminary results have been obtained. The present paper summarizes recent results from those studies.
Sogabe, Toshiaki; Ishihara, Masahiro; Baba, Shinichi; Tachibana, Yukio; Yamaji, Masatoshi*; Iyoku, Tatsuo; Hoshiya, Taiji*
Materials Science Research International, 9(3), p.235 - 241, 2003/09
2D-C/C composite is one of the promising materials as a next-generation core material in gas-cooled reactors. Effect of air-oxidation on the thermal diffusivity up to 1673K of the 2D-C/C composite was investigated. The C/C composite specimens for measurement of thermal diffusivity were oxidized from 1 to 11 percent weight loss in air at 823K. Thermal diffusivity at room temperature declined 10 20 percent for parallel to lamina direction and 5
20 percent for parallel to lamina direction and 5 9 percent for that of perpendicular within 11 percent weight loss by oxidation. Thermal diffusivity tended to decrease gradually as the increase of oxidation loss in parallel to lamina, however, it decreased in the beginning of oxidation pretty much and not so changed by further oxidation loss in perpendicular to lamina. Change in thermal conductivity under oxidation condition was also estimated.
9 percent for that of perpendicular within 11 percent weight loss by oxidation. Thermal diffusivity tended to decrease gradually as the increase of oxidation loss in parallel to lamina, however, it decreased in the beginning of oxidation pretty much and not so changed by further oxidation loss in perpendicular to lamina. Change in thermal conductivity under oxidation condition was also estimated.
Yoshigoe, Akitaka; Moritani, Kosuke; Teraoka, Yuden
Japanese Journal of Applied Physics, Part 1, 42(7B), p.4676 - 4679, 2003/07
Times Cited Count:1 Percentile:5.63(Physics, Applied)Many studies of the thermal oxidation on Si(001) surface by O gas have been already carried out from both experimental and theoretical methods. The oxidation reaction kinetics have been studied by real time photoemission spectroscopy. Most reports, however, were performed at a fixed electron binding energy. We present the study of initial stage of thermal oxidation on Si(001) surface at the O
gas have been already carried out from both experimental and theoretical methods. The oxidation reaction kinetics have been studied by real time photoemission spectroscopy. Most reports, however, were performed at a fixed electron binding energy. We present the study of initial stage of thermal oxidation on Si(001) surface at the O pressure of 1x10
pressure of 1x10 Pa performed by real time O-1s synchrotron radiation photoemission spectroscopy.All experiments were performed at SUREAC2000 at BL23SU in the SPring-8. The pure O
Pa performed by real time O-1s synchrotron radiation photoemission spectroscopy.All experiments were performed at SUREAC2000 at BL23SU in the SPring-8. The pure O gas of 1x10
gas of 1x10 Pa was fed into the reaction analysis chember through a variable leak valve. The results of oxygen uptake curves obtained by O-1s peak area intensities at the substrate temperature of 855K and 955K indicate that the Langmuir adsorption model provided the best fitting result for the 855K oxidation, whereas the oxidation at 955K was well explained by the autocatalytic reaction model.
Pa was fed into the reaction analysis chember through a variable leak valve. The results of oxygen uptake curves obtained by O-1s peak area intensities at the substrate temperature of 855K and 955K indicate that the Langmuir adsorption model provided the best fitting result for the 855K oxidation, whereas the oxidation at 955K was well explained by the autocatalytic reaction model.
 gas on Si(0 0 1) surface by means of synchrotron radiation Si-2p photoemission spectroscopy
gas on Si(0 0 1) surface by means of synchrotron radiation Si-2p photoemission spectroscopyYoshigoe, Akitaka; Moritani, Kosuke; Teraoka, Yuden
Applied Surface Science, 216(1-4), p.388 - 394, 2003/06
Times Cited Count:9 Percentile:45.93(Chemistry, Physical)It is well known that the initial Si(0 0 1) oxidation by O gas is an important reaction system because it is usually used to form gate-oxide films on MOSFET. With decreasing the size of ULSI, it is necessary to control the surface reaction with atomic scales. In this study, we report
gas is an important reaction system because it is usually used to form gate-oxide films on MOSFET. With decreasing the size of ULSI, it is necessary to control the surface reaction with atomic scales. In this study, we report 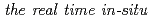 observation of thermal oxidation using O
observation of thermal oxidation using O gas on Si(0 0 1) surface by means of synchrotron radiation photoemission spectroscopy at the soft x-ray beamline, BL23SU, in the SPring-8. We clarified the chemisorption processes of O
gas on Si(0 0 1) surface by means of synchrotron radiation photoemission spectroscopy at the soft x-ray beamline, BL23SU, in the SPring-8. We clarified the chemisorption processes of O on Si(0 0 1) surface over 773K regions at the initial oxidation stages from the results of Si2p core-level shifts. The fundamental understanding of surface reaction is expected to contribute the development of the future nanotechnology.
on Si(0 0 1) surface over 773K regions at the initial oxidation stages from the results of Si2p core-level shifts. The fundamental understanding of surface reaction is expected to contribute the development of the future nanotechnology.
Yoshigoe, Akitaka; Moritani, Kosuke; Teraoka, Yuden
Japanese Journal of Applied Physics, Part 1, 42(6B), p.3976 - 3982, 2003/06
Times Cited Count:6 Percentile:28.14(Physics, Applied)It is well known that the thermal oxidation on Si(001) surface is an important reaction system to form of the gate-oxide films in MOSFET, since it is necessary to control the film thickness under a few nano-meter scale. Thus, we have studied the oxygen uptake and the Si oxidation states depending on the oxidation times by using the synchrotron radiation photoemission spectroscopy in 1 10
10 Pa of O
Pa of O at the surface temperature from 870K to 1120K. We clarified the oxidation depending on the surface temperature was explained by the kinetics (Langumuir and auto-catalytic model). Using real time photoemission spectroscopy, we found that the Si
at the surface temperature from 870K to 1120K. We clarified the oxidation depending on the surface temperature was explained by the kinetics (Langumuir and auto-catalytic model). Using real time photoemission spectroscopy, we found that the Si sepcies was not formed at the initial oxidation stage.
sepcies was not formed at the initial oxidation stage.
Hirota, Koichi; Arai, Hidehiko; Hashimoto, Shoji
Bulletin of the Chemical Society of Japan, 73(12), p.2719 - 2724, 2000/12
Times Cited Count:7 Percentile:38.34(Chemistry, Multidisciplinary)no abstracts in English
Fujii, Kimio; Yamada, Reiji
Journal of Nuclear Materials, 258-263, p.1953 - 1959, 1998/00
Times Cited Count:8 Percentile:57.22(Materials Science, Multidisciplinary)no abstracts in English
*; Hirata, Masaru; Yahata, Taneaki
Journal of Nuclear Science and Technology, 28(3), p.228 - 238, 1991/03
no abstracts in English
Seguchi, Tadao; Morita, Yosuke; Yoshida, Kenzo
EIM-84-131, p.27 - 36, 1984/00
no abstracts in English
; ;
JAERI-M 83-070, 49 Pages, 1983/05
no abstracts in English
; ;
Journal of Nuclear Science and Technology, 20(11), p.941 - 950, 1983/00
Times Cited Count:35 Percentile:93.84(Nuclear Science & Technology)no abstracts in English
Seguchi, Tadao; Arakawa, Kazuo
EIM-81-91, p.11 - 20, 1981/00
no abstracts in English
*; *
Bulletin of the Chemical Society of Japan, 37(2), p.244 - 248, 1964/00
Times Cited Count:11no abstracts in English
*; *
Bulletin of the Chemical Society of Japan, 36(6), p.717 - 721, 1963/00
Times Cited Count:16no abstracts in English
*; *; *; Terakawa, Akira*
Bulletin of the Chemical Society of Japan, 36(6), p.721 - 727, 1963/00
Times Cited Count:20no abstracts in English